Q4B Evaluation and Recommendation of Pharmacopoeial Texts for Use in the ICH Regions Annex 6 Uniformity of Dosage Units General Chapter
ICH guideline Q4B annex 6 to note for evaluation and recommendation of pharmacopoeial texts for use in the ICH regions on unifor

PDF) Weight and content uniformity of lorazepam half-tablets: A study of correlation of a low drug content product
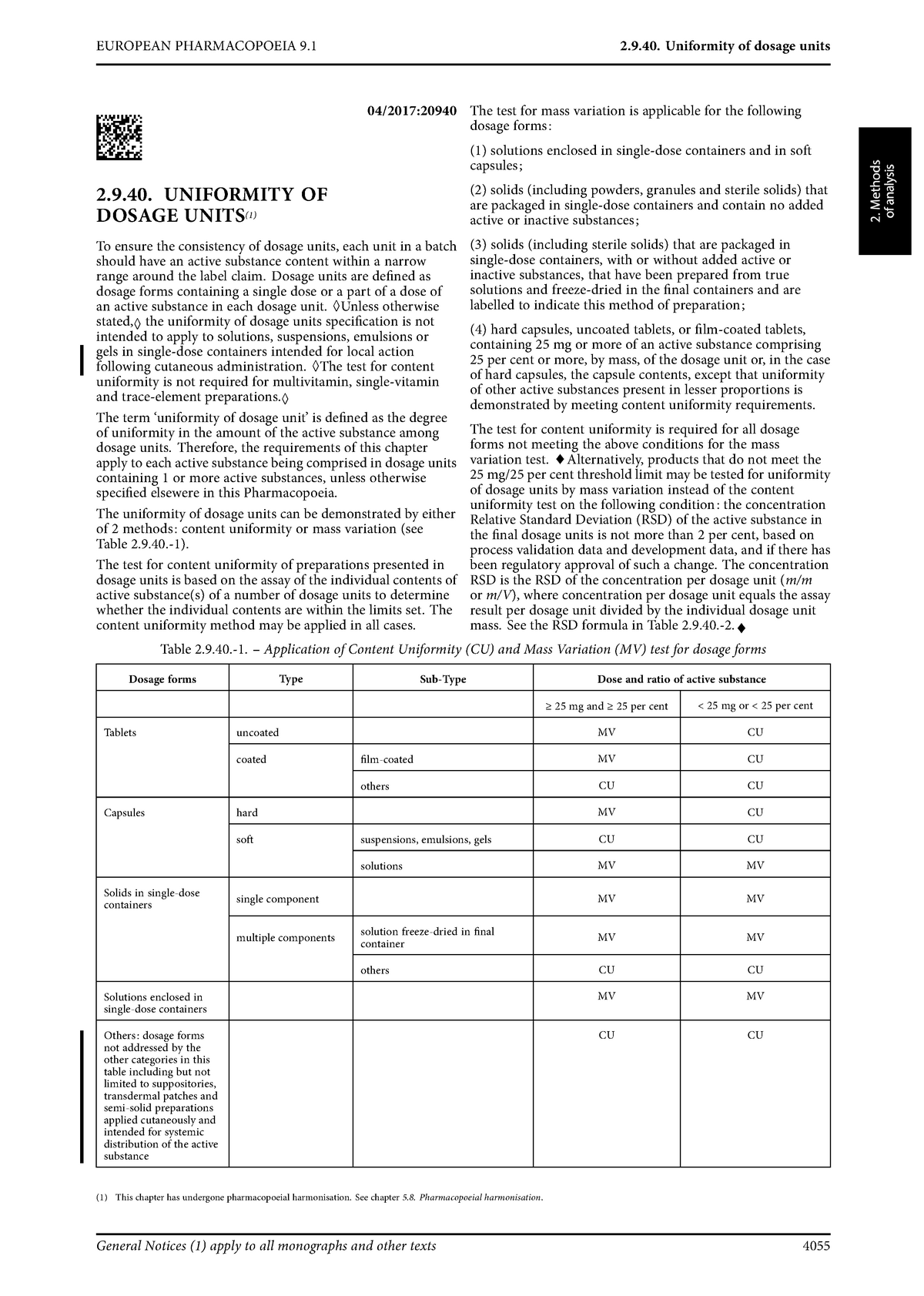
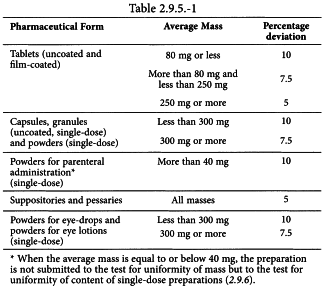
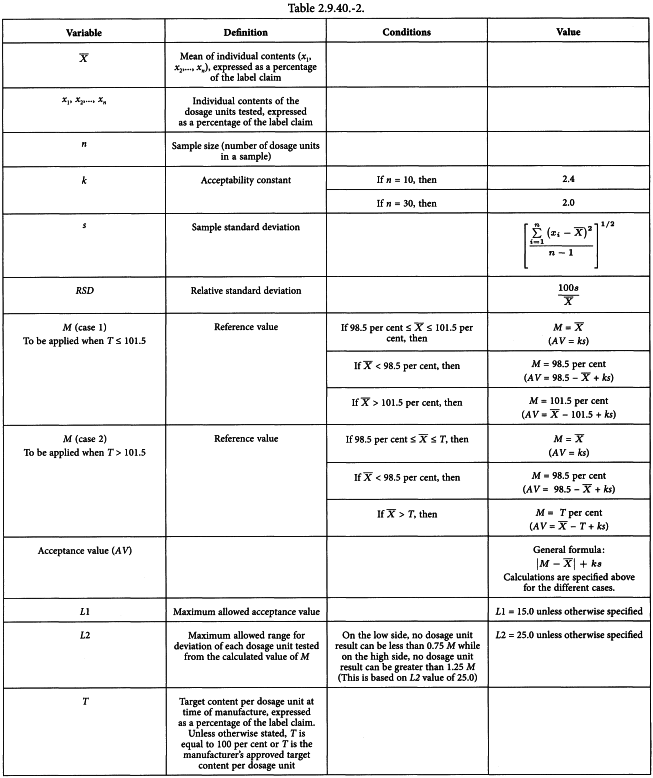
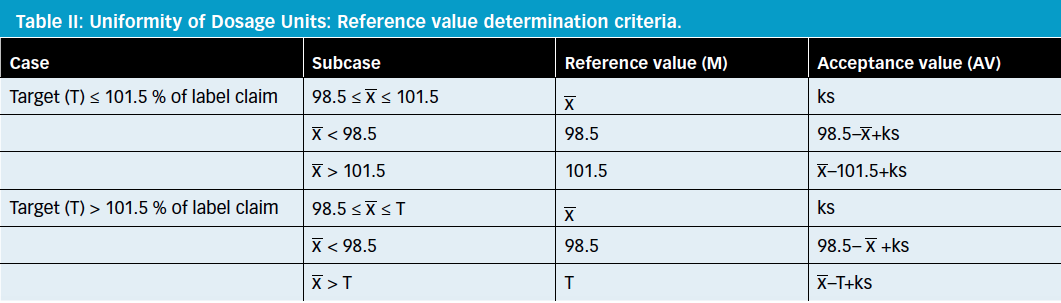
Uniformity of dosage units - EUROPEAN PHARMACOPOEIA 9 2.9. Uniformity of dosage units 04/2017: 2.9. - Studocu

Uniformity of dosage units—comparative study of methods and specifications between Eur. Pharm. 3rd and USP 23 - ScienceDirect

ASTM E2810-19 - Standard Practice for Demonstrating Capability to Comply with the Test for Uniformity of Dosage Units

PDF) Weight and content uniformity of lorazepam half-tablets: A study of correlation of a low drug content product

Uniformity of dosage units—comparative study of methods and specifications between Eur. Pharm. 3rd and USP 23 - ScienceDirect